Breaking Update: Here’s a clear explanation of the latest developments related to Breaking News:Real-World Analysis of the ImmunoWELL JCV Test in Natalizumab ▼ Progressive Multifocal Leukoencephalopathy Risk Monitoring– What Just Happened and why it matters right now.
Introduction
JCV is a DNA polyomavirus and the primary aetiological agent of PML.1,2 Most adults carry JCV in its latent form, but the virus can become reactivated during treatment with immunosuppressive monoclonal antibodies such as natalizumab.1,2 Serum anti-JCV antibody testing is recommended prior to initiating natalizumab therapy, with retesting every 6 months due to considerations such as new infection or fluctuating antibody levels.3,4
The ImmunoWELL JCV test is currently used in the clinical practice setting in Europe to support PML risk stratification in patients with MS receiving or considering natalizumab.5-7 This test is a validated, two-step, enzyme-linked, immunosorbent assay that qualitatively detects anti-JCV antibodies in human serum.5-7 It utilises a screening assay that measures antibody–antigen binding with peroxidase-conjugated anti-human IgG antibodies and categorises the sample as anti-JCV antibody ‘positive’, ‘negative’, or ‘indeterminate’. If the sample reports an indeterminate result, a confirmation test on the same sample (second step) is required.7
European Real-World Analysis of the ImmunoWELL JCV Test
Esmael et al.8 conducted the first European real-world analysis of the ImmunoWELL JCV test for PML risk monitoring. In this study, aggregated ImmunoWELL JCV test data from April 2024–April 2025 were retrospectively analysed across eight European countries: France, Germany, Italy, the Netherlands, Norway, Spain, Sweden, and the UK. Testing was performed by Medicover Integrated Clinical Services (MICS; Gdańsk, Poland) and its affiliated laboratory in Bucharest, Romania.8
Samples were categorised as ‘new’ or ‘re-tests’ based on prior anti-JCV antibody testing history. Analyses included the monthly distribution of ImmunoWELL JCV test volumes, anti-JCV antibody status using qualitative assessment based on index values (≤0.20 negative and >0.50 positive), and PML risk stratification based on the following index threshold values: low ≤0.8, moderate >0.8–≤1.4, and high >1.4.8
ImmunoWELL JCV Test Data
A total of 16,763 test results were analysed between April 2024–2025, comprising 77.3% (12,965/16,763) new and 22.7% (3,798/16,763) re-tests. Of the total number of tests, 39.1% (6,550/16,763) were anti-JCV antibody-negative, and 60.9% (10,213/16,763) were anti-JCV antibody-positive (Figure 1).8
Figure 1: Percentage distribution of anti-JCV antibody-positive and -negative samples normalised by country (N=16,763).8
JCV: John Cunningham virus.
When considering the distribution of samples across PML risk index categories (low, moderate, high), most results fell into the low-risk category. The overall test distribution for low (index: ≤0.8), moderate (index: >0.8–≤1.4), and high (index: >1.4) PML risk was 79.7% (13,360/16,763), 7.4% (1,245/16,763), and 12.9% (2,158/16,763), respectively. Norway reported the highest percentage of tests in the low PML risk category (88.2%), whereas the UK recorded the highest percentage in the high PML risk category (19.5%; Figure 2).8
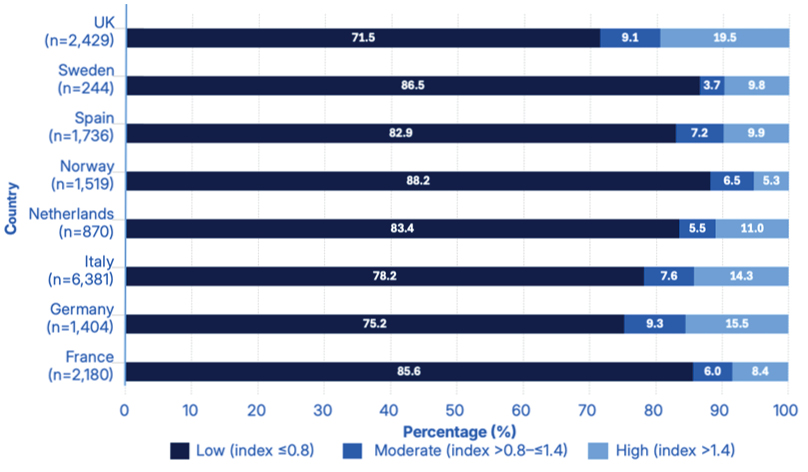
Figure 2: Percentage distribution of samples across progressive multifocal leukoencephalopathy risk index categories (low, moderate, high) normalised by country (N=16,763).8
The ‘low PML risk’ category includes all test results with an index value of ≤0.8, regardless of antibody status. Further analysis of the distribution of anti-JCV antibody-positive and -negative samples in the low PML risk category was therefore carried out.
Overall, 47.7% (6,373/13,360) of samples within this category were anti-JCV antibody-positive, while 52.3% (6,987/13,360) were anti-JCV antibody-negative (Figure 3).8 The observation that nearly half of the samples within the low risk group were positive for anti‑JCV antibodies further supports the high sensitivity of the ImmunoWELL JCV assay.9
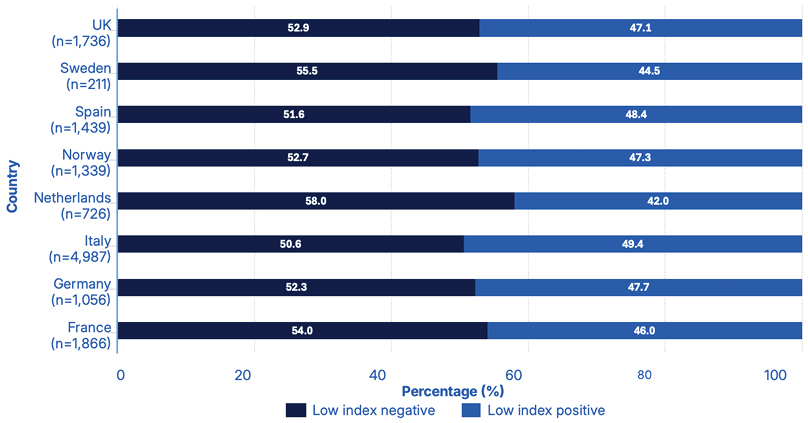
Figure 3: Percentage distribution of anti-JCV antibody-positive and -negative samples in the low progressive multifocal leukoencephalopathy risk category (index ≤0.8), normalised by country (N=13,360).8
JCV: John Cunningham virus.
Conclusion
Evaluation of the distribution of anti-JCV antibody index values obtained using the ImmunoWELL JCV test, with over 16,700 results collected from eight countries over the course of 1 year, found that approximately 80% fell within the low PML risk category (≤0.8).8 This indicates a predominance of low-risk profiles among tested individuals, as well as a consistent pattern of results across different European countries. Importantly, the overall percentage reported for low PML risk using the ImmunoWELL JCV test was broadly consistent with current known practices in clinical risk assessment and treatment decision-making.10
In summary, this first multinational, real-world analysis of the ImmunoWELL JCV test supports its clinical utility as a reliable tool for anti-JCV antibody assessment and monitoring in patients with MS receiving or considering treatment with natalizumab.

