Breaking Update: Here’s a clear explanation of the latest developments related to Breaking News:Microbubbles – Works in Progress Magazine– What Just Happened and why it matters right now.
One of the central problems in medicine is delivering drugs in the body, at the right time, place, and concentration. In many cases, less than one percent of an injected cancer drug dose actually reaches the tumor. The body is difficult terrain to navigate and unforgiving to outsiders. Some drugs need to evade immune cells, and many fail due to unsuccessful delivery. But the brain is even more forbidding. It has a defensive barrier that excludes nearly all large drugs, such as antibody therapies and nanoparticles, and most small molecule drugs, such as most chemotherapy drugs. This makes it much harder to treat conditions like epilepsy, Alzheimer’s and Parkinson’s than diseases in the rest of the body.
To overcome these problems, researchers have long experimented with vessels that could transport drugs to their destination while shielding them from the body’s defense system. These include nanoparticles, which are tiny structures made from metals, polymers or lipids that are about a thousandth of the width of a single human hair; liposomes, which are fatty spherical pouches whose walls are made from the same material as cell membranes; and nanobots, hypothetical miniature machines that could perform tasks at the molecular or cellular level.
Get the print magazine
Subscribe for $100 to receive six beautiful issues per year.
Subscribe
But all of these face challenges. The liver and spleen intercept a large fraction of nanoparticles before they ever reach the target (though they still show promise for certain breast and lung cancers, as they can more easily permeate blood vessels). Liposomes face a similar problem: macrophages in the liver recognize and engulf most of them on the journey. Working nanobots are still a distant prospect. And most are blocked from reaching the brain. These are some of the barriers that microbubbles may be able to cross.
Boy in the bubble
Microbubbles are just what they sound like: tiny gas-filled bubbles. Scientists have engineered them further, to be coated with a protective outer shell and made capable of carrying drugs or genetic material to cells in the body. They are microscopic, roughly the width of spider silk, but still hundreds or thousands of times larger than nanoparticles or liposomes. This means they’re too large to leave the bloodstream. Instead, microbubbles deliver drugs by bursting on command.
As they burst open, they briefly force open biological barriers that are otherwise impenetrable, such as the blood-brain barrier, allowing treatments to pass through. The force of their bursts can even be the treatment itself, as they could also be used to break apart kidney stones.
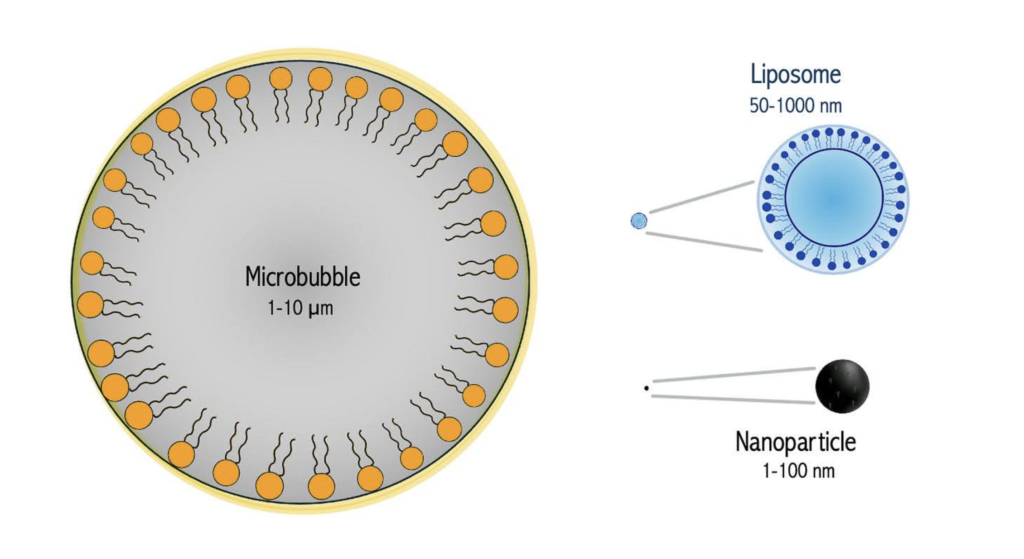
Microbubbles are far larger than liposomes or nanoparticles.
Microbubbles were first developed to help radiologists read scans. In the late 1960s, doctors at the University of Rochester were using ultrasound to take pictures of heart structures when they made an accidental discovery. When they injected saline into a vein near the heart, their ultrasound image lit up with a cloud of bright signals. The flashes had been created by tiny air bubbles, formed by the rush of fluid from the injection: sound waves passed through blood and tissue fairly smoothly, but once they encountered gas, the change in density reflected the waves back to the sensor.
That discovery became a standard technique for spotting structural defects in the heart: if bubbles crossed from one chamber to the other on the scan, it indicated a hole. But the bubbles were short-lived and inconsistent in size, so researchers began experimenting with other injectable substances, including blood and medical dyes, to produce stronger, more stable signals. These efforts to engineer reliable contrast agents for ultrasound imaging mark the beginning of what we now call medical microbubbles.
Over the following decades, researchers moved from experimenting with whatever was at hand to deliberately designing microbubbles from scratch, giving them thin shells of lipids, proteins, or polymers to control their size and keep them stable long enough to be clinically useful.
One approach to create a protective shell was to use albumin, which is familiar as the protein in egg whites but is also the most common protein in the bloodstream, where it is used to ferry molecules through the body. Since the body already produces it in large quantities, it doesn’t provoke an immune response and can be used medically. It also has useful structural properties: it unfolds and hardens into a shell sturdy enough to survive circulation, but is brittle enough that it can fall apart when hit by focused sound waves. Albunex, an albumin-coated and air-filled microsphere, was approved by the FDA in the early 1990s for use in cardiac ultrasounds, specifically to make the inner walls of the heart’s left ventricle visible on a scan. Unlike air bubbles, these albumin-coated microbubbles were stable with refrigeration for at least two years.
Researchers also found they could make even sturdier shells through sonication: blasting a protein-rich liquid with high-energy sound waves. The intense energy causes the protein molecules to assemble spontaneously with their water-repelling sides facing inward toward the gas, and their water-loving sides facing out, and as they do, the acoustic energy chemically cross-links them in place, forming a rigid shell. Unfortunately, air inside dissolves readily into the surrounding blood, causing the bubbles to collapse within seconds. Scientists found they could dramatically extend bubble lifetime by replacing the air with heavier, less soluble gases, such as perfluorocarbons and sulfur hexafluoride, which linger inside the shell long enough to be clinically useful. These second-generation microbubbles can persist in the bloodstream for several minutes, and can meaningfully enhance ultrasound images of the heart and liver.
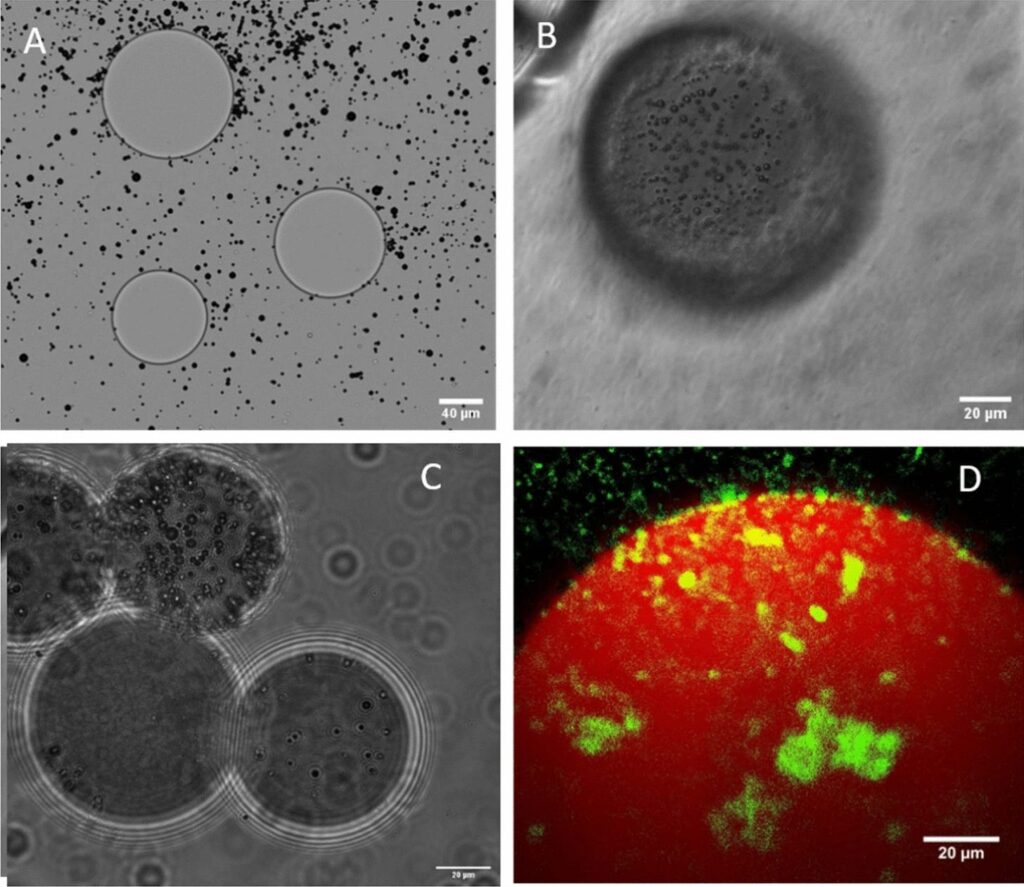
Microbubbles (smaller circles) on the surface of beads (larger circles).
The cruise missiles of medicine
Microbubbles, being bubbles, have many other interesting properties beyond reflecting sound and carrying material: they can expand, contract and burst.
This behavior, known as cavitation, was already familiar to physicists: when a bubble collapses, it generates a brief pressure pulse that can push on surrounding material. Lawrence Crum, a researcher from the University of Washington, had shown this in larger bubbles, but the question was whether the same forces could be harnessed usefully in living tissue.
A group at Stanford found that they could. At lower intensities, cavitation didn’t destroy cells. Instead, it temporarily loosened the walls of blood vessels and punched small, reversible pores in cell membranes, in a process called sonoporation. This opened a new possibility for drug delivery, especially in the brain, where the blood-brain barrier tightly regulates what can enter. Sonoporation offered a way to open that barrier temporarily and locally, letting drugs through without permanently disrupting it.
Bubbles also move with what surrounds them: wind, water, blood. In the bloodstream, that would usually mean being swept along by the current, with no control over where they end up. Scientists from Northwestern hypothesized that by steering the bubbles, they could control the drugs. They eventually cracked it in the early 1980s by attaching magnetic nanoparticles to microbubble shells made of albumin and treating rats with tail tumors by injecting microbubbles into a nearby artery and using a magnet to capture the bubbles at the tumor site.
A microbubble future
Microbubbles have been used in diagnostics since the 1990s, but haven’t yet been approved for delivering medical treatments. Part of the reason comes down to how they’re tested. Imaging microbubbles, which are already in clinical use, are relatively straightforward to evaluate. Therapeutic bubbles are harder, because their success depends on both whether the bubbles reach their target and whether the treatment they carry actually works. The external ultrasound system adds another layer of complexity, as researchers need to make sure the ultrasound pulses are working correctly and reliably too. Imaging bubbles also had a head start: they were developed first, and built up a safety record in thousands of patients through large multicenter trials in the 1990s, while therapeutic microbubbles are still catching up.
But researchers are exploring their use across a wide range of conditions: dissolving blood clots in stroke patients, delivering chemotherapy directly to tumors, opening the blood-brain barrier for brain cancer treatments, and even targeting genetic material like mRNA to specific tissues.
Microbubbles are different from many other delivery platforms because they can be steered. But some use cases are more promising than others: treatments where the target is physically accessible, such as a tumor with a clear boundary or a clot in a vessel, are better suited to microbubbles than diffuse conditions where the problem is spread across many tissues.
Brain cancer is likely one promising area: microbubbles can open the blood-brain barrier and early trials in glioblastoma patients have shown the approach is feasible with manageable side effects such as passing headaches. In one small pilot trial, the cavitation effect from bursting bubbles was used to oxygenate tumors, which are otherwise harder to treat with radiation.
Pancreatic cancer is another: in a small 2016 pilot trial, patients treated with microbubble-delivered gemcitabine survived on average eight months longer than the control group, and tolerated more treatment cycles. These are small trials, and results like these need to be replicated at scale.
The same idea is likely to extend beyond cancer. To treat ischemic strokes, doctors currently deliver a clot-dissolving protein called tPA intravenously, but because it circulates through the whole body, it also interferes with healthy clotting elsewhere. Microbubbles could release tPA directly at the clot site, avoiding that tradeoff. Similarly, they also offer a way to deliver chemotherapy drugs like doxorubicin and gemcitabine directly to tumors, sparing the heart, liver, and bone marrow from the damage that comes with high systemic doses. And for mRNA therapies, including vaccines like those developed against Covid-19, current delivery systems distribute the vaccines across many tissues, wasting much of it.
Kidney stones are another option, as a physical object that could be broken apart with microbubbles. Researchers have proposed equipping microbubbles with chemical tags that bind specifically to the mineral in most kidney stones, then triggering cavitation with an ultrasound pulse to burst them from inside. Focused ultrasound devices for kidney stones have already received FDA clearance, which suggests regulators are comfortable with the broader approach.
Most drug delivery is systemic: drugs circulate through the entire body, and only a fraction reaches the intended site; the rest is either wasted or can cause off-target side effects. Microbubbles, on the other hand, could carry a therapy through the bloodstream and release it precisely where it’s needed, guided by an external ultrasound pulse. It turns out that to solve some of medicine’s unsolved problems, you sometimes need to think very, very small, and then burst through.

