Breaking Update: Here’s a clear explanation of the latest developments related to Breaking News:Expert Perspectives on the Use of Subcutaneous Infusion Therapies in Parkinson’s Disease– What Just Happened and why it matters right now.
Interview Summary
Subcutaneous (SC) infusion therapy with apomorphine first became available for the treatment of Parkinson’s disease (PD) in Europe in the early 1990s. SC infusion options have expanded in recent years to include levodopa-based formulations, and were introduced in the USA in 2024. This article collates expert insights from PD specialists in Europe and the USA, with extensive cumulative experience of using SC infusion therapies to treat their patients, to address knowledge gaps and share best practice with the wider neurology community. To this end, EMJ conducted interviews between December 2025–January 2026 with nine key opinion leaders: Rajesh Pahwa, University of Kansas Medical Center, Kansas City, USA; Michael Soileau, Texas Movement Disorder Specialists, PLLC, Georgetown, USA; Stuart Isaacson, Parkinson’s Disease and Movement Disorders Center of Boca Raton, Florida, USA; Robert Hauser, University of South Florida, Tampa, USA; Angelo Antonini, University of Padua, Italy; Tove Henriksen, University Hospital of Bispebjerg, Copenhagen, Denmark; Regina Katzenschlager, Department of Neurology and Karl Landsteiner Institute for Neuroimmunological and Neurodegenerative Disorders, Klinik Donaustadt, Vienna, Austria; and Andrew Lees and Cathy Magee from the National Hospital for Neurology and Neurosurgery, London, UK. Based on their insights, this article discusses the SC infusions available for PD and clinical evidence for their efficacy and safety; when and for which patients they should be considered; and how to optimise their use in real-world practice, including proactive skin care and reliable, user-friendly delivery systems.
INTRODUCTION
PD is the second most common neurodegenerative disease,1 characterised by bradykinesia plus rest tremor or rigidity.2 The discovery of levodopa for the treatment of PD revolutionised its management. However, while oral levodopa-based treatment regimens provide symptom control in the short-to-medium term for most patients, long-term levodopa treatment is frequently associated with the eventual development of motor fluctuations and dyskinesia.3 Motor fluctuations can occur as early as the first 1–2 years of treatment with levodopa in some patients, by 5 years in approximately 50% of patients, and by 10 years of treatment, most patients experience OFF episodes.4
The management of motor fluctuations typically involves adjusting the patient’s levodopa regimen by altering the dose and/or dosing frequency or switching to an extended-release formulation; adding an oral or transdermal dopamine agonist (if not already used); adding adjunctive medication such as a catechol-O-methyltransferase or monoamine oxidase-B inhibitor to slow the breakdown of levodopa or dopamine, respectively; or using non-dopaminergic levodopa extenders such as adenosine A2A antagonists or amantadine. However, treatment response can remain unpredictable, especially with advancing disease, and on-demand rescue medication is often required. One factor that contributes to loss of efficacy of oral levodopa is poor absorption due to gastrointestinal (GI) dysfunction, which is a common feature of PD.5,6 It is therefore rational to consider non-oral routes of administration. SC infusion not only bypasses the GI tract, but also provides continuous drug delivery, and, therefore, continuous dopaminergic stimulation, avoiding the peaks and troughs associated with oral medication.
Until recently, in the USA, progressing beyond oral/transdermal medications as the core treatment regimen has largely required surgical procedures to place a deep brain stimulation (DBS) device or jejunostomy tube for intrajejunal delivery of carbidopa/levodopa enteral suspension (CLES), usually referred to as levodopa/carbidopa intestinal gel in Europe. The recent introduction of treatments administered via SC infusion, apomorphine and foscarbidopa/foslevodopa (fosCD/fosLD), provides an alternative therapeutic strategy or a potential intermediate step in the treatment pathway before advancing to surgical options. However, many healthcare professionals in the USA are not yet familiar with the available SC options. There is greater familiarity in Europe, where SC apomorphine has been available for >30 years (with formulations available for delivery by injection or as a continuous SC apomorphine infusion [CSAI]), and SC fosCD/fosLD (infusion solution) was launched in 2024.7
Although CSAI has been available in Europe for several decades, Lees commented that it has tended to be underused.8 The expansion of SC treatment options to include fosCD/fosLD, coupled with their wider availability across regions, makes this an opportune time to examine when and how SC infusion therapies can be used to improve the management of PD. This article brings together the expertise and experience of PD specialists in both Europe and the USA to share their collective knowledge regarding SC infusion as a treatment modality for PD with the wider neurology community.
OVERVIEW OF SC INFUSION THERAPIES FOR PD: CLINICAL EVIDENCE
There are currently two approved products (CSAI and fosCD/fosLD) for the treatment of PD via SC infusion. Clinical evidence for these treatments has been reviewed elsewhere;9 key efficacy and safety data from clinical trials are summarised briefly below.
Apomorphine (Continuous SC Apomorphine Infusion )
The most robust evidence for the efficacy and safety of CSAI comes from the TOLEDO study, a double-blind, randomised, placebo-controlled trial in 106 patients with PD and persistent motor fluctuations, despite optimised oral or transdermal medication.10 After 12 weeks’ double-blind treatment, CSAI reduced mean OFF time by almost 2 hours per day more than placebo (treatment difference: −1.89 hours per day; p=0.0025).10 Reductions in OFF time were sustained throughout a 52-week open-label phase, coupled with increased ON time without troublesome dyskinesia and reductions in mean oral levodopa-equivalent dosage.11 The most common treatment-emergent adverse events (AE) included infusion site reactions (e.g., nodules/erythema), nausea, somnolence, and dyskinesia.10 Six patients (11%) discontinued treatment during the 12-week double blind treatment period for various reasons, including one discontinuation due to infusion site reaction. By the end of the open-label phase, 17% of patients had discontinued CSAI, including four patients (5%) discontinuing due to infusion site reactions.
The InfusON study (NCT02339064) was a 52-week open-label study of CSAI in 99 patients enrolled at 19 centres in the USA, which supported its approval in the USA.12 Results were consistent with those of the TOLEDO study, with mean OFF time reduced by about 3 hours by Week 12, and a corresponding increase in ON time without troublesome dyskinesia and reduction of oral levodopa-equivalent dosage and adjunctive medication use. Improvements were sustained through Week 52, and the majority of patients reported their symptoms were much or very much improved at Weeks 12 and 52. The tolerability profile was consistent with expected AEs based on TOLEDO and clinical experience in Europe.12
Foscarbidopa/Foslevodopa
Foscarbidopa and foslevodopa are carbidopa and levodopa prodrugs. The efficacy and safety of fosCD/fosLD for SC infusion were investigated in a 12-week double-blind RCT (NCT04380142)13 and a 52-week open-label study (NCT03781167).14 In the 12-week RCT (N=141), ON time without troublesome dyskinesia increased by a mean of 2.72 hours with fosCD/fosLD versus 0.97 hours with oral carbidopa/levodopa (difference: 1.75 hours; p=0.0083), with a similar reduction in OFF time.13 Infusion site events were common (72% with fosCD/fosLD versus 12% with SC placebo), including erythema, pain, oedema, nodules, and cellulitis; these largely accounted for a higher discontinuation rate (22% versus 1%). Other AEs included dyskinesia, falls, and hallucinations.13
In the 1-year open-label study, all patients (N=244) received fosCD/fosLD, with dosage optimised during the first 4 weeks of treatment.14 Mean ON time without troublesome dyskinesia increased by 3.8 hours from baseline to Week 52, and mean OFF time decreased by 3.5 hours.14 Interim analysis of a long-term extension study indicates that efficacy was sustained in patients who opted to continue treatment for up to 96 weeks.15 Similar to the shorter-term study, the most common AEs in the 52-week open-label study were infusion site reactions, including erythema, nodules, cellulitis, and oedema.14 A total of 107 patients (44%) discontinued treatment within the 1-year study period, including 56 (23%) whose primary reason for discontinuation was AEs. The most common AEs leading to discontinuation were hallucinations, dyskinesia, and infusion site reactions, including erythema, cellulitis and nodules.14 The authors suggested that “the prevention and management of infusion site AEs should be among the top priorities to consider when initiating fosCD/fosLD to promote adherence to therapy.”14 Expert contributors discuss skin management later in this article.
The ongoing ROSSINI study (NCT06107426) is a real-world observational study investigating the long-term safety and tolerability of fosCD/fosLD.16 An interim analysis found that patients treated with fosCD/fosLD for 6 months (N=105) had statistically significant reductions in mean OFF time (−2.7 hours) and dyskinesia time (−1.7 hours). Improvements were also observed in Movement Disorders Society Unified PD Rating Scale (MDS-UPDRS) Part III scores and measures of sleep, pain, quality of life, freezing of gait, and GI dysfunction.17
THE PLACE OF SC INFUSION THERAPY IN THE PD TREATMENT PATHWAY
When Should Physicians Consider Introducing SC Infusion Therapies?
Antonini outlined the ‘5-2-1’ criteria as a useful rule-of-thumb used in Europe for recognising patients with advanced PD who might benefit from stepping up treatment. The 5-2-1 rule refers to patients who require oral levodopa dosing ≥5 times daily, or have ≥2 hours per day with OFF symptoms, or have ≥1 hour per day of troublesome dyskinesia.18,19 Antonini explained that these simplified criteria, which are easier to remember than a complex algorithm, are a useful guide for general neurologists to identify patients with advanced PD who may need to be referred to a specialist centre for the next phase of treatment.
Meeting any one of the 5-2-1 criteria is considered sufficient to prompt consideration of device-aided therapies, including SC infusion.18,19 This does not set an excessively high threshold for considering advanced therapy, and Antonini supported early introduction of SC infusion therapy. He cited a post-hoc analysis of the fosCD/fosLD RCT, focusing on a subgroup of younger patients (≤65 years) at an earlier stage of advanced PD than the overall study population (≤5 years since onset of motor fluctuations; disease duration <10 years in >90% of patients [n=26]). For most efficacy endpoints, treatment benefit of fosCD/fosLD in this subgroup was consistent with, and numerically greater than, the overall study population.20 This finding is important, Antonini said, because “it shows that the earlier you use this treatment the better your [outcomes] are.” Lees has longstanding experience with CSAI and welcomed a trend towards its earlier use. In the past, it was unusual to introduce CSAI until approximately 9–10 years after diagnosis, whereas 6–7 years post diagnosis is now more typical.
In practice, it is not just the numerical criteria, but their functional impact that indicates a need to progress beyond oral therapy. Prompts to consider advanced treatment may include loss of independence due to unmanageable fluctuations and difficulty maintaining compliance with oral medication regimes. Patients may be taking levodopa 4–5 times per day and are often advised to avoid taking it close to mealtimes to limit protein interactions, which can affect absorption, making it difficult to integrate medication schedules into daily life. Soileau explained that patients reach a point where the therapeutic window for oral levodopa is very narrow, with a limited dose range that provides adequate relief of motor symptoms without inducing dyskinesia. At this point it becomes difficult to manage symptoms using oral medication, presenting “a good opportunity to try SC therapy.”
Which Patients Are Suitable Candidates for SC Infusion Therapy Versus DBS?
Core eligibility criteria are similar for SC infusion therapy and DBS: a patient experiencing 2–3 hours’ OFF time per day, with OFF episodes causing some functional difficulty, who has tried at least one adjunctive therapy in addition to levodopa could be a candidate for either SC infusion or DBS. Cognitive status might determine treatment selection, as DBS is not suitable for patients with significant cognitive impairment. For SC infusion therapy, patients need to have capacity to operate the pump and infusion set or have support from a caregiver if they cannot manage this themselves, which is not precluded by mild/moderate cognitive impairment. Patients with severe dementia are not considered suitable candidates for infusion therapy (SC or intrajejunal) due to the risk that, if a patient becomes confused by the presence of the infusion equipment, they might pull out the cannula or disconnect the pump.
For patients who are eligible for either option, the choice is usually determined by patient preference, with physician guidance. Henriksen emphasised the importance of involving patients in treatment decisions, as having the patient ‘on board’ with the treatment selected has been shown to improve compliance. Different treatment options should be presented neutrally, explaining pros and cons of each option without bias, to allow patients to make informed treatment decisions.
The experts interviewed for this article listed several advantages of SC infusion therapy: it is less invasive than surgical options and it is also reversible. If a patient has difficulty managing the pump or does not tolerate SC treatment well, they can simply stop treatment and would still be candidates for DBS. Another advantage is that SC infusions can be started quickly in the clinic, whereas DBS requires extensive assessments and neurosurgical consultation to confirm suitability before the surgery itself can be scheduled. Several experts talked about using SC infusion as a ‘bridging therapy’ for patients awaiting DBS.21 Given the non-invasive and reversible nature of SC infusion therapies, there was broad agreement among the experts that it is usually preferable to try SC infusion before DBS. Antonini described SC infusion as a “good entry point” to device-aided therapy.
The main reason given for some patients expressing a preference for DBS over SC infusion was not wanting to carry an infusion pump around. Additionally, once the DBS device is implanted, patients do not have to think about it, whereas SC infusion requires daily management and skin care.
Table 1 summarises some of the key considerations that might influence selection between SC infusion versus DBS for patients with advanced PD. Anecdotally, experts suggested that use of CLES, another advanced option that requires both surgery and a pump for drug delivery, appears to have decreased slightly with the availability of fosCD/fosLD as an SC option for continuous infusion of levodopa-based treatment. Selection between DBS, CLES, and CSAI is reviewed elsewhere.22 Results from a network meta-analysis suggested potentially greater OFF time reduction with DBS and CLES than with CSAI.23 However, no direct head-to-head comparative studies have been performed. The network meta-analysis23 pre-dated approval of fosCD/fosLD, so evaluation of SC infusion therapy was limited to CSAI.
Table 1: Factors that may influence choice of SC infusion versus DBS.
*The pump can be removed for showering/bathing using the disconnect feature on the NeriaTM Guard
(Convatec, London, UK) infusion set.
DBS: deep brain stimulation; SC: subcutaneous.
It is not necessarily an either/or choice between different device-aided therapies, but a question of which option to try first. Patients who have undergone SC infusion therapy often subsequently move on to DBS.24 Conversely, patients can have SC infusion therapy added to DBS if they continue to experience OFF episodes or motor fluctuations. As Soileau put it: “Patients can try SC therapy at any point in their journey; having more options sends a very encouraging message to the patient and care-partner.”
Overall, the option of SC infusion therapy may broaden the pool of patients who could benefit from device-aided therapy, as the threshold for consideration of this reversible option may be lower than for more invasive device-aided therapies,9 and some patients for whom DBS would be contraindicated due to cognitive impairment could be eligible for SC therapy. Soileau welcomed the availability of more treatment options (following introduction of SC infusion therapies in the USA), noting that having different options allows physicians to tailor treatment to a patient’s specific OFF symptoms. For example, DBS might be more suitable than fosCD/fosLD for a patient with levodopa-resistant tremor, whereas freezing gait can be worsened with DBS and CSAI might be more beneficial.
A proposed schema for positioning SC infusion therapies in the treatment pathway is shown in Figure 1. It is important to note that there is no single preferred pathway, and an individualised approach is required to meet patients’ specific needs.
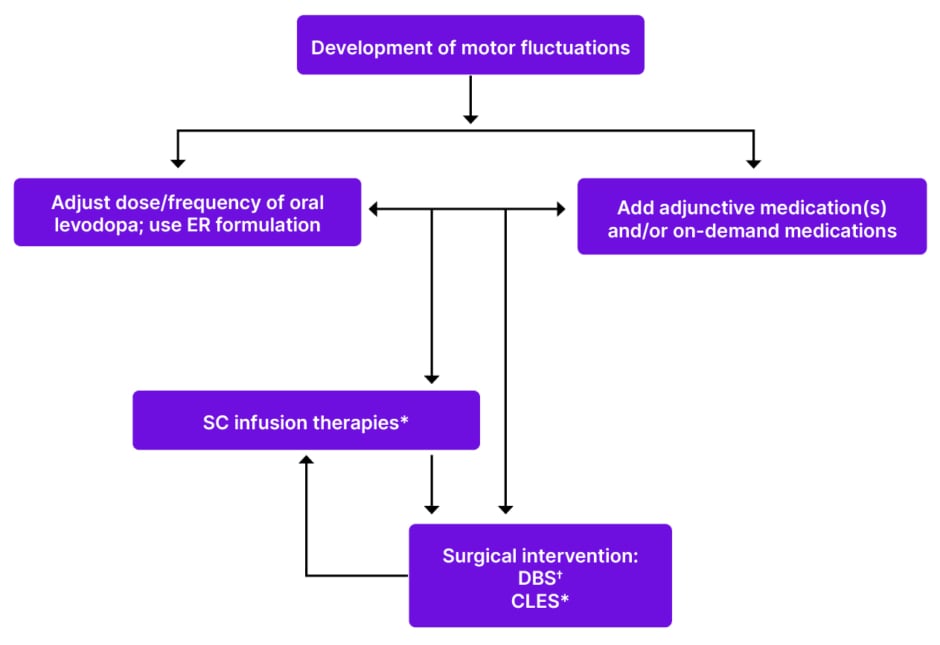
Figure 1: Proposed placement of SC infusion therapies in the PD treatment pathway.
*Candidates for infusion therapies should have capacity or support to manage infusion equipment and should not have significant dementia.
†Candidates for DBS should not have significant cognitive impairment.
Patients with OFF time ≥2 hours with some functional difficulty who have tried ≥1 adjunctive medication are eligible for device-aided therapy.
Proposed positioning does not account for approval/reimbursement status of different treatment options in regional healthcare systems, which may dictate eligibility for specific options.
CLES: carbidopa/levodopa enteral solution; DBS: deep brain stimulation; ER: extended release; SC: subcutaneous;
PD: Parkinson’s disease.
What Factors Influence Treatment Selection Between CSAI or Foscarbidopa/Foslevodopa?
Once SC infusion has been selected as the preferred therapeutic approach, the next decision is between CSAI or fosCD/fosLD. This must be considered on an individual basis for each patient. No head-to-head studies have directly compared the efficacy and safety of CSAI and fosCD/fosLD. Lees’ group conducted an indirect treatment comparison based on published literature, which suggested comparable efficacy of the two treatments.25 Safety profiles were also broadly similar, though there was a trend towards better tolerability of CSAI, driven by higher rates of infusion site reactions and hallucinations with fosCD/fosLD, which is borne out in real-world clinical experience (as discussed later in this article). Since magnitude of effect is similar across different SC infusion therapies, treatment choice will come down to other factors.
Cost may be one factor;25 however, cost–benefit considerations and reimbursement arrangements for each treatment in different regions are beyond the scope of this article. Based on clinical considerations only, factors that might influence treatment choice include: preference for monotherapy versus adjunctive treatment; infusion duration; prior response to oral levodopa or dopamine agonists (patients who have responded well to oral levodopa, but lost efficacy due to declining GI absorption, are likely to respond to levodopa-based treatment via other routes of administration, while good response to oral/transdermal dopamine agonists might predict good response to CSAI); predisposition to side effects (also based on prior experience with oral levodopa-based medication or dopamine agonists); symptom profile (for example, CSAI can help non-motor symptoms, including apathy); and pump characteristics such as size, weight, and complexity.
The “beauty of having options,” Soileau said, is that you can start one SC treatment and monitor the patient’s response, and, if the response is unsatisfactory, there is still another SC option to try. Switching SC therapies is relatively easy for the patient as the same infusion set can be used, so their routine of changing needles, and associated steps, would not need to change. Although the pumps differ, patients are familiar with the concept of using a pump for continuous drug delivery.
PRACTICAL MANAGEMENT OF PD USING SC INFUSION THERAPIES: REAL-WORLD CLINICAL EXPERIENCE
This article reflects the perspectives of experts practicing in several European countries and the USA, revealing differences in some aspects of practical management that are dictated by local health systems. Local logistical considerations are not addressed in detail here. This article focusses on clinical aspects of management, on which there was a high level of agreement between contributors.
Before Initiation
Acceptance of treatment can be improved by proactively discussing the concept of continuous infusion with patients at an early stage in the disease course. Alongside communicating benefits (Antonini explained how he talks to patients about being able to achieve a more constant clinical state and simplifying their treatment regimen), it is important to set realistic expectations. Magee commented that patients need to be aware that SC treatment is “not a panacea,” and must be prepared for practical aspects of care, including managing the infusion equipment and maintaining good skin management.
Where available, a prophylactic anti-emetic (domperidone) may be administered before starting CSAI. Magee indicated that this is common practice in the UK; however, Pahwa noted that domperidone is not available in the USA and nausea is mitigated through slow titration from a low starting dose.
Initiation and Titration
SC infusion therapy may be initiated in outpatient clinics, in the hospital inpatient setting, or at home; approaches varied between practitioners in different countries, as did the level and nature of nursing support available. Several European experts highlighted benefits of admitting patients as inpatients for a few days to facilitate close monitoring and support during initiation and titration. Lees explained that hands-on support can help patients get over initial ‘teething problems’ with infusion equipment. Specialist centres often take patients referred from a wide area and admit patients for whom it is not practical to travel to the clinic for daily assessments and adjustments during the titration phase. However, inpatient admission is not the best approach for all patients; for example, some patients with neurocognitive deficits may become disoriented by the unfamiliar environment. Magee noted that the COVID-19 pandemic forced a move towards more home-based care, even by specialist centres that had typically provided inpatient care in the UK, by nurses with specialist training under close supervision to guide titration. This approach has been retained where possible for patients who prefer home-based care.
In the USA, SC treatment is initiated in outpatient clinics or at home. The ability to manage patients in the outpatient setting was viewed as an advantage over more invasive treatments that require costly admission for surgery. At the present time, fosCD/fosLD is usually initiated and optimised in day clinics, while CSAI can be initiated at home, in the clinic, or using a hybrid approach.
Initiation of CSAI is usually performed in the ON state, as infusion is started at a low dose and can take several hours to take effect. Recommendations for initiating CSAI have been published elsewhere.26,27 Briefly, a typical initiation protocol begins at a low dose (1 mg/hr), with dosage increased in small increments (0.5 mg/hr) to a maximum of 6 mg/hr. The speed of the titration depends on the setting and on toleration of treatment. In some European centres, inpatients with excellent tolerability may achieve a steady dose in around 7–10 days under close monitoring. In outpatient and home settings, titration usually proceeds less rapidly over several weeks.26 If the patient was using an oral or transdermal dopamine agonist prior to starting CSAI, this is gradually reduced as it is replaced by CSAI during the titration window to avoid dopamine withdrawal syndrome. Concomitant levodopa is also tapered down. CSAI is generally used as an adjunct to oral levodopa, but a key therapeutic goal is to reduce oral levodopa dosage as far as possible to minimise dyskinesia and reduce the burden of oral medication regimens,9 and some patients ultimately achieve monotherapy with CSAI.
For fosCD/fosLD, the starting dose is guided by the patient’s existing oral levodopa dosage, aiming to replace oral levodopa with an equivalent dose of fosCD/fosLD. A conversion formula that adjusts for molecular weight and bioavailability of foslevodopa versus levodopa is provided in the prescribing information28 and via an online calculator. However, Katzenschlager and Antonini cautioned that this does not account for incomplete GI absorption of oral levodopa. Many patients with impaired GI absorption take high nominal oral dosages to achieve an effective therapeutic dose. As the proportion of the oral dose that ultimately reaches the brain cannot be reliably determined, direct conversion from nominal oral dosage may risk overestimation. Infusion can be started at a lower dose than that calculated based on oral dosage and titrated to find the therapeutic window that reduces OFF time without inducing dyskinesia. Concomitant dopamine agonists can be withdrawn, ultimately aiming for fosCD/fosLD monotherapy, but should be tapered down gradually, not stopped abruptly, to reduce the risk of dopamine agonist withdrawal syndrome.
Optimising Infusion Duration and Flow Rate
CSAI is usually administered for approximately 16 hours per day, with 8 hours’ break overnight, while fosCD/fosLD is intended for continuous 24-hour infusion. In practice, however, duration can be varied to reflect individual motor patterns, non‑motor symptoms, and tolerability.
While some patients may continue CSAI throughout the day and night, this constitutes off-label use. Some centres use transdermal or long-acting oral dopamine agonists to provide dopaminergic treatment during the night for patients stopping infusion overnight. Patients may experience morning OFF episodes after an overnight break from CSAI and require a bolus dose on waking. The infusion pump has an ‘extra dose’ facility to administer a loading dose on restarting infusion in the morning. However, cannulating and reconnecting the pump may be challenging for patients with motor impairment on waking (unless assistance is provided by a caregiver), so some patients instead administer a dose of SC apomorphine using an injection pen, which typically takes effect within approximately 5–15 minutes, or use alternative on-demand medication. Isaacson advised establishing a stable dosage for continuous infusion before enabling the extra dose function.
Although primarily intended for daytime use, Lees outlined an alternative use of CSAI in patients with nighttime symptoms. He described the APOMORPHEE study, a double-blind, placebo-controlled crossover study in 46 patients with PD and moderate-to-severe insomnia, in which nighttime-only CSAI was associated with significant improvement in mean PD sleep scale scores, compared with placebo.29
FosCD/fosLD is now available for patients requiring 24-hour dosing to manage nighttime symptoms, provide motor control overnight (for example, for patients who need to mobilise to use the bathroom at night), or avoid morning OFF episodes. In Pahwa’s experience, most patients welcome the benefits of 24-hour dosing, but some patients on fosCD/fosLD do break from treatment overnight; for example, if they find it more comfortable to remove the pump to sleep or as a measure to manage side effects. Neuropsychiatric AEs, particularly hallucinations, can manifest as nightmares, and in such cases discontinuing treatment overnight can be beneficial. However, Henriksen noted that patients who interrupt treatment overnight can experience a prolonged morning OFF period, and for this reason, overnight discontinuation is rarely recommended at her centre. Instead, the experts interviewed generally advocated reducing the infusion rate to administer a lower dose overnight.
The fosCD/fosLD pump has base, low, and high flow-rate settings that patients can switch between. The infusion rates delivered at each setting can be adjusted according to clinical need. Antonini suggested setting the low rate at approximately 50% of the base rate to provide substantially lower overnight dosing, whereas Hauser reported using low and high rates set 10% below and above the base rate, respectively. Antonini cautioned against frequent day-to-day changes in flow rate, recommending a simple regimen of base rate during the day and low rate overnight. This approach facilitates interpretation of treatment response to guide dose adjustments at follow‑up visits. The pump also includes an ‘extra dose’ function to deliver bolus doses if OFF symptoms emerge, which can be useful for patients experiencing morning OFF symptoms after reduced overnight dosing. However, bolus doses take 2–3 hours to provide benefit, so other on-demand medications with faster onset, such as levodopa inhalation powder or dispersible tablets, may be required.
Using the Infusion Set
A crucial element of the initiation visit, whether in the clinic or at home, is educating the patient and their caregivers on correct use of the infusion kit. Patients with preserved manual dexterity are often able to manage infusion-set changes independently, although in some cases assistance from a relative or caregiver is required either routinely or during periods of impaired motor function.
Many centres use an SC infusion set with an automated insertion device to standardise technique and reduce needlestick risk. All contributors to this article reported using the NeriaTM Guard (Convatec, London, UK) infusion set with both CSAI and fosCD/fosLD. This system comprises a soft cannula with a retractable introducer needle, which is secured to the body at the chosen infusion site using an adhesive patch, an insertion device that deploys the needle into the skin, and tubing that connects the cannula to the pump (Figure 2). The introducer needle remains hidden from the patient throughout the process of insertion using the automated device, which may make SC infusion more accessible to patients with needle phobia.
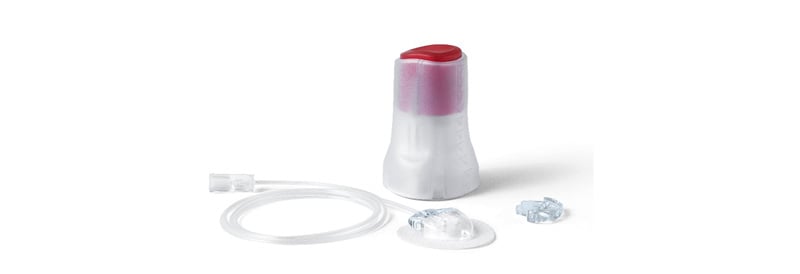
Figure 2: Infusion set for use with continuous subcutaneous apomorphine infusion or subcutaneous foscarbidopa/foslevodopa pump.
NeriaTM Guard (Convatec, London, UK) soft cannula infusion set with an integrated insertion device.
Key considerations for optimal use include:
- Frequent changing: Infusion sets should be changed daily for most patients, as prolonged use could increase risk of infusion site reactions. Although instructions for fosCD/fosLD recommend changing the infusion set every 72 hours, there was broad agreement among the experts interviewed for this article that it is best practice to change infusion sets more frequently to maintain cleanliness and allow regular repositioning. Most advised patients to change sets daily initially, with the option to extend to every other day if patients tolerate infusions well. Since CSAI is discontinued overnight, a new infusion set is used when the infusion is restarted each day.
- Site selection: A new site should be used each time the infusion set is changed, with systematic rotation to allow adequate healing. Suitable sites with sufficient SC adipose tissue include the abdomen, flanks, thighs, and upper arms. Although abdominal sites close to the umbilicus are recommended in fosCD/fosLD manufacturer instructions,28 this restriction limits site rotation. Using sites over a wider body area allows patients to rotate between sites they find most comfortable and does not affect fosCD/fosLD bioavailability.30
- Hygiene: Rigorous hygiene is essential to minimise infection risk. Patients are taught aseptic technique, including handwashing and cleaning the site before applying the adhesive patch. Soileau emphasised the importance of teaching patients and caregivers good hygiene practices, saying “educate, educate, educate,” while Magee concurred that the importance of hygiene “cannot be stressed enough.”
- Cannula insertion: The skin should be pulled taut with the insertion device held flush against the skin so that the needle enters perpendicular to the surface. Full penetration of the dermis is required to ensure delivery into the SC space. The NeriaTM Guard infusion set can be used with a 9 mm or 6 mm cannula; all contributors to this article expressed a preference for the 9 mm cannula to ensure complete penetration of the dermis. The automated insertion device makes it easier for patients to achieve the correct insertion angle and depth compared with older infusion sets that required manual insertion of the cannula/needle.
- Connecting and operating the pump: Tubing should be connected to the pump and primed with solution before attachment to the cannula to avoid introducing air. While infusion rates are programmed by healthcare staff, patients must learn to operate user-controlled features, such as low/high flow rate selection for fosCD/fosLD and extra doses.
- Post-insertion: Gentle massage of the infusion site after starting the infusion, for example, using a handheld massager, helps disperse medication to reduce nodule formation.
Troubleshooting: Pain, burning, or visible pooling of medication beneath the skin suggests inadequate needle penetration; in such cases, patients should immediately re-cannulate, using a new infusion set rather than attempting to reposition the existing needle. Infusion solution ‘oozing’ onto the skin also suggests incorrect insertion, as the cannula is primed with medication only after deployment, during automated insertion with NeriaTM Guard, to prevent the solution from coming into contact with the skin surface. Liquid should be wiped away promptly to avoid irritation.
From a practical perspective, infusion-set design and human factors can be critical success factors for SC therapy.31 For example, automated insertion simplifies training and supports reliable drug delivery by improving consistency of cannula placement, and reduced needle handling improves safety. Magee described such innovations as “game-changing,” noting that ease-of-use can mean “the difference between infusion therapy working and not working.”
Follow-Up
Experts consistently highlighted the importance of providing support in the early stages of SC infusion therapy to help establish treatment successfully and reduce the risk of discontinuation. Follow-up visits, conducted at home or in the clinic, provide an opportunity to troubleshoot problems with the infusion set or pump and to monitor side effects. Patients should also be able to access support, ideally on a 24-hour basis, to permit prompt reporting of issues that arise, particularly skin reactions.
Managing Adverse Reactions
Infusion site reactions are a potential concern with any infused medication. Skin reactions including nodules, erythema, pain, and cellulitis have been reported with SC infusion therapies for PD.10-14 Effective mitigation of such reactions is important to promote treatment adherence. Good hygiene and frequent changing and repositioning of infusion needles are key steps. Isaacson also suggested that decreasing the infusion flow rate can help ensure medication is absorbed and does not accumulate in the SC space and form nodules, alongside a gentle massage to aid dispersion.
Distinguishing between inflammatory reactions and infection at the infusion site can be challenging in clinical practice. Redness around the site could indicate either; several interviewees recommended taking photographs and marking the margins of erythema to help monitor changes in size and colour. Heat or pus may indicate infection. Doxycycline, which has anti-inflammatory as well as antibiotic properties, can be prescribed if infection is suspected. Topical corticosteroids can be used to alleviate local inflammation when infection is not suspected.
European experts with longstanding experience with CSAI commented that they had observed a decline in rates of skin problems over the years, owing to a combination of growing experience of managing AEs and improvements in infusion set/needle technology. FosCD/fosLD, however, is associated with a higher rate of skin complications, including infections and abscesses, which can impact treatment adherence. Magee observed that repeated infections requiring antibiotics had contributed to the decision to discontinue fosCD/fosLD for some patients in her clinic. Specific strategies to manage fosCD/fosLD-related skin reactions may emerge as clinical experience accumulates.
Aside from skin reactions relating to the mode of administration, patients may experience side effects associated with dopaminergic medication, including neuropsychiatric adverse events, such as hallucinations or psychosis, and nausea. Titrating up slowly from a low starting dose is recommended to reduce the risk of these AEs. Orthostatic hypotension, which is commonly associated with oral dopamine agonists and injectable apomorphine, appears to be less common with CSAI, Soileau observed, possibly reflecting a “gentler, smoother” drug concentration profile.
Advances in SC Infusion Therapy
There have been significant advances in SC infusion technology since CSAI first became available. Speaking based on several decades of experience with CSAI, Lees explained that pumps have become smaller and more ambulatory, and infusion sets have evolved substantially. Modern infusion sets such as NeriaTM Guard provide standardised insertion and reduced needle handling, and contributors highlighted the availability of the 9 mm cannula option as particularly useful to ensure consistent SC delivery. Lees noted that current needle systems with soft polytetrafluoroethylene cannulas are less irritative than older infusion sets with steel needles, and discontinuation of CSAI because of skin nodules is now uncommon in his clinic. Henriksen has also observed reduced rates of skin problems since introducing NeriaTM Guard compared with older infusion devices that were available when she began using CSAI in her clinic approximately 25 years ago. Overall, Henriksen perceived CSAI as a well-tolerated therapy based on her extensive clinical experience.
As fosCD/fosLD has only recently entered real-world clinical practice, a learning curve for prescribing clinicians is anticipated,32,33 particularly with respect to identifying and managing practical challenges. In real-world settings, clinicians are already adapting manufacturer guidance, for example by changing infusion sets more frequently (daily instead of every 72 hours) and rotating infusion sites across wider body regions, to mitigate risk of skin reactions. Magee emphasised the importance of providing clear instructions to patients regarding these adaptions, as discrepancies between advice from the treating clinician and the manufacturer’s instructions may otherwise lead to confusion.
CONCLUSION
The addition of SC infusion therapies to the treatment armamentarium for advanced PD expands the range of options available to patients who experience motor fluctuations on oral levodopa-based regimens and provides a potential alternative to surgical interventions. Greater choice to select the best treatment to meet patients’ needs was welcomed by experts, particularly in the USA, where SC infusion therapy is a relatively new option. Treatment success with SC infusion therapy is improved by proactive mitigation and effective management of adverse effects, particularly infusion site reactions, to minimise avoidable treatment discontinuation. The overall ‘therapy system’ offers opportunities to further optimise treatment outcomes in real-world practice through dose individualisation, structured patient and caregiver education, and reliable delivery via user-friendly infusion equipment.

